Instruments
Filter
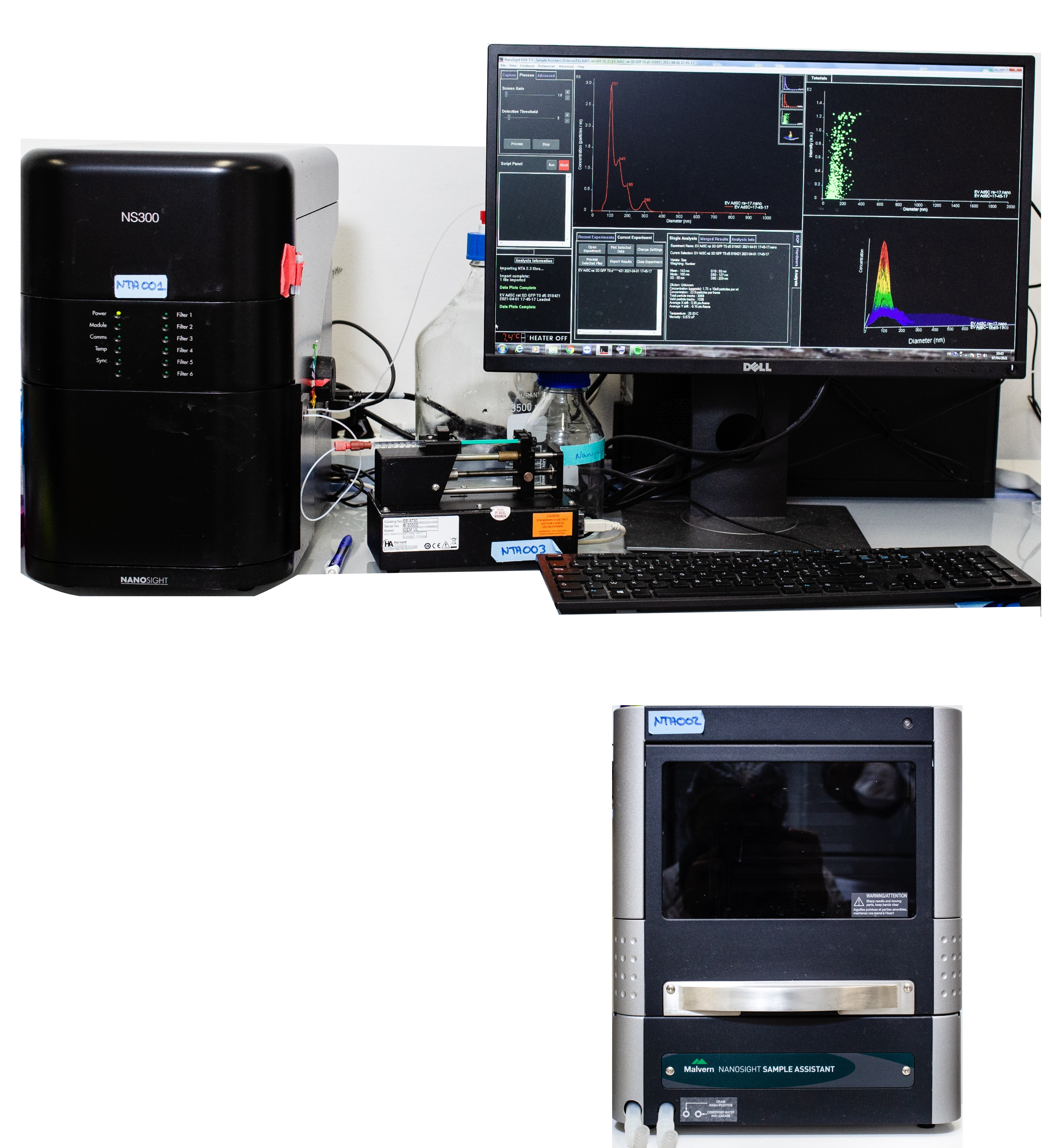
Nanosight NS300 – Malvern Panalytical
Nanosight NS300 allows the characterization of nano-object’s size and concentration. It is a laser-based technique where particles are detected following light scattering. The trajectory of several particles is followed and registered by a ccd camera and the hydrodynamic radius is deduced from the Stokes-Einstein equation governing Brownian motion trajectories of spherical particles in pure viscous medium. Nanosight NS300 is also equipped with a Nanosight syringe pump and nanosight sample assistant for a single or multiple samples, respectively.
Specifications:
Excitation wavelength: 405 nm,
Optimal concentration range: 1 – 5*108 part/mL,
Optimal size range: extracellular vesicles: 50 – 1000 nm, metallic nanoparticles: down to 10 nm,
Rate: ~ 6 min / sample (5*60s protocol)
Tracked particles: 250
https://www.malvernpanalytical.com/en/products/product-range/nanosight-range/nanosight-ns300
Nanosight syringe pump (single sample)
Nanosight NS300 is equipped with a syringe pump (1mL syringe) allowing measurement to be done at constant flow rate therefore improving sample statistics.
Specifications:
Optimal sample volume: 1000 µL
Nanosight Sample Assistant (multiple samples)
Nanosight Sample Assistant is an autosampler enabling automatic loading of multiple samples up to 96.
Specifications:
Support: 96 well plate
Optimal sample volume: 1mL / well
Rate (example): 16 hours for 96 samples with a 3*60s protocol
VideoDrop - Myriade
VideoDrop is a particle analyzing device used to perform physical characterization (size and concentration) of particles of 80-400nm size. Particles are illuminated with a monochromatic (450 nm) light source and their detection is based on interferometric light microscopy (ILM). ILM is a new innovative method allowing to measure live (< 1min) concentration and size of particle in a single drop (few μL) of a sample. Videodrop detects the interference patterns by the means of the light scattered by particles. Then, based on the counted particles in a defined volume the concentration is identified, while particles’ size (median hydrodynamic diameter) is deduced from their trajectories due to Brownian motion through the Stokes-Einstein equation.
Specifications:
Excitation wavelength: 450 nm,
Sample volume: 5-10 µL
Optimal concentration range: 108 – 1010 part/mL,
Optimal size range: extracellular vesicles: 80 – 500 nm, metallic nanoparticles: down to 30 nm,
Rate: ~ 1 min / sample
Tracked particles: 100 - 500

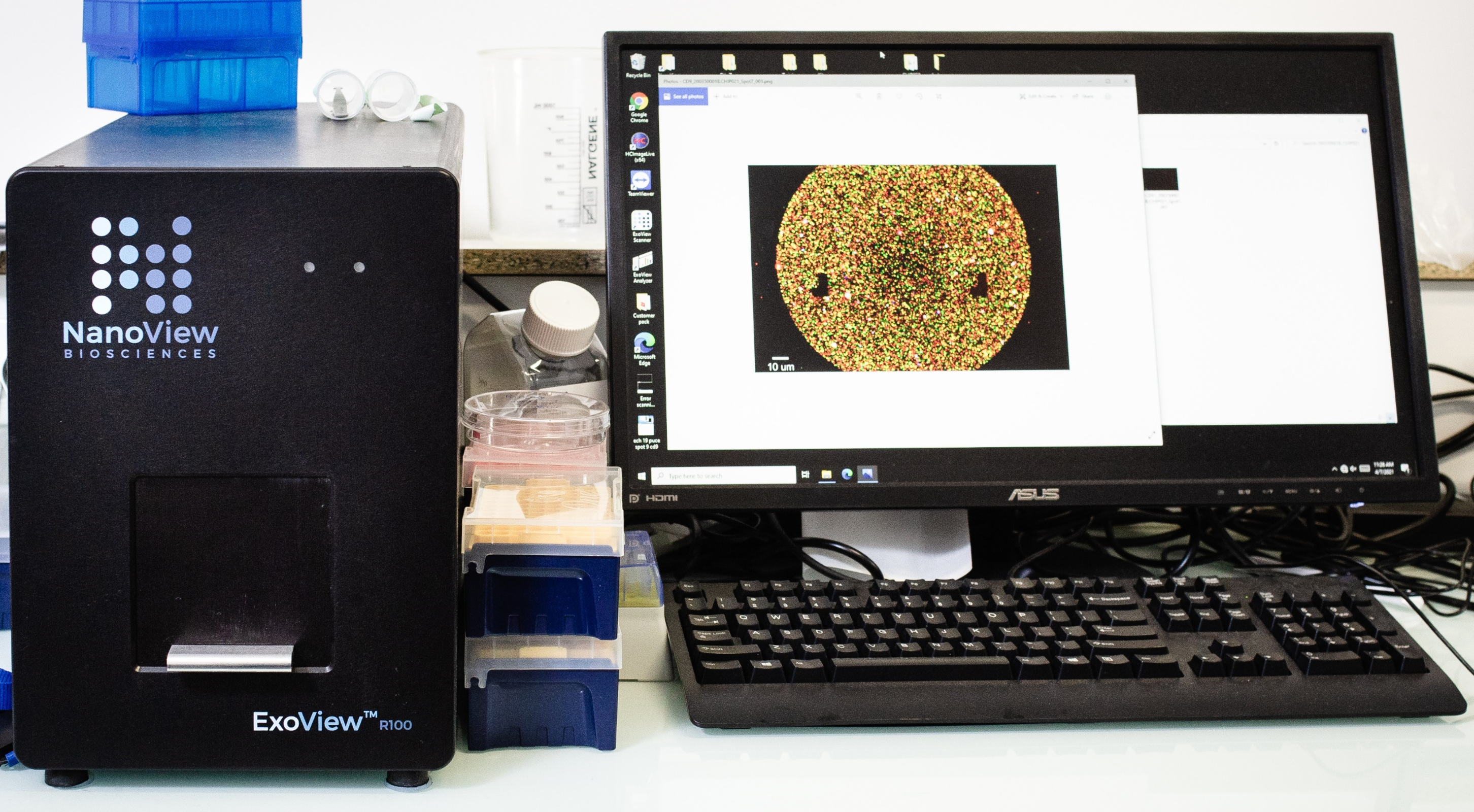
ExoView R100 – NanoView
The ExoView® R100 device from Nanoview is an interferometric and fluorescence detection system for the relative quantification and analysis of the size of extracellular vesicles (EVs). EVs attach to a microchip (from the Nanoview supplier) functionalized with the CD9, CD63, CD81 (EV markers) and IgG antibodies (negative control). Chips with other antibodies can be custom ordered. Detection fluorescent antibodies can be chosen according to the needs of the experimenter enabling to investigate the co-localization up to 4 markers on EVs. Color channels, excitation and emission filters are indicated below:
| Specifications | ||||
| Color | Filter of excitation | Filter of emission | ||
| Blue (488) | 480 +/- 15 nm | 519 +/- 13 nm | ||
| Green (555) | 555 +/- 12.5 nm | 595 +/- 16.5 nm | ||
| Red (647) | 640 +/- 15 nm | 697 +/- 30 nm | ||
EVs can be detected even in the presence of contaminants such as protein aggregates (the discrimination being made by the absence of binding between the contaminants and the capture antibodies present in the microchip).
The system also allows the study of the co-localization of membrane and cytosolic markers (located inside EVs). Cytosolic markers are detected after a step of fixation and permeabilization before the addition of the fluorescent antibody with affinity by the cytosolic marker of interest, either positive or negative.
The system allows the analysis of 9 chips at one run (about 10 min per chip).
Chips can be prepared in your own lab and transported for reading at the IVETh core facility.
https://www.nanoviewbio.com/products/exosome-detection/exoview-r100-automated-exosome-measurement
Nucleocounter® NC-200™ – Chemometec
The NucleoCounter® NC-200™ is an automated device for cell count and viability measurements. The analysis is performed using a Via1-Cassette™ (from the Chemometec supplier) for automated and standardized staining and sample handling. The cassette comprises two fluorescent dyes pre-loaded: acridine orange (AO) that stains all cells green and DAPI that stains dead cells blue. The disposable cassette is volume-calibrated to ensure high-precision counts and cell reproducibility. An integrated image analysis algorithm acquires and analyzes a fluorescent image of the cells, recording their intensity and size.
Besides the analysis of cells in suspension (detached adherent cells or non-adherent cells), measurements can be performed for cell aggregates and cells on microcarriers by performing a cell disruption step. A dedicated protocol is operational (2 steps, via two reactants from Chemometec supplier), but adaptation may be necessary in a case-by-case basis.
The NucleoView™ software features a module that allows implementation of electronic record keeping, electronic signatures, and administrative rights to maintain paperless record systems under good manufacturing practice (GMP) guidelines.
Specifications:
Detection range: 5*104 – 5*106 Cells/mL
Minimal volume: 100µl (loading volume: 60µl)
Rate: 50s (1 step protocol) – 120s (2 steps protocol)
https://chemometec.com/products/nucleocounter-nc-200-automated-cell-counter/


Asymetrical flow field – flow fractionation (A4F) – Wyatt
Eclipse™ module for FFF
Eclipse™ module for FFF
Field-flow fractionation is a wide dynamic range, versatile means of size-based separations, capable of fractionating biotherapeutics such as viral particles, liposomes and nanoparticles, emulsions, proteins and aggregates, extracellular vesicles with no stationary phase and almost no shear. The separation can be performed in the formulation buffer. The A4F system is driven by an Agilent Infinity II G7111B HPLC which includes: an autosampler, a UV-Vis spectrometer, a fluorescence detector, a fraction collector and a thermostating unit.
General specifications:
LC NEON® and Dispersion Neon® channel.
DCM Module.
Spacer 125, 190, 250, 350 et 490 μm.
Membrane PES or RC, 10kDa.
Size: 2nm – 800 nm
Flow rate: 0.1-8.3mL/min
https://www.wyatt.com/products/instruments/eclipse-field-flow-fractionation-system.html
DAWN™ module for MALS (18 angles)
DAWN™ module for MALS (18 angles)
The DAWN® is a Multi-Angle static Light Scattering (MALS) detector for absolute characterization of the molar mass and size of biomolecules and nanoparticles in solution. When combined with a size based separation method and a quantitative detection (such as refractometry and UV spectrometer), the MALS detection allows to determine the molar weights and the gyration (or geometric) radius. When combined with multiple detections (A4F-MALS-DLS-UV-Fluo-RI), the analysis allows to relate the size distribution of biomolecules and nanomedicine to other information (content, biomarkers, morphology).
Specifications:
Molar Mass Range: 200 Da to 1 GDa.
Molecular Size Range (Rg) : 10 to 500 nm, up to 1000 nm with shape-specific models
Molecular Size Range (Rh) : In flow mode, 0.5 nm to 300 nm2; batch mode 0.5 nm to 1 μm
Sensitivity 10 ng or 100 kDa for polystyrene in THF or 200 ng of BSA in PBS
Laser Wavelength 658 nm ± 4 nm
https://www.wyatt.com/products/instruments/dawn-multi-angle-light-scattering-detector.html
OPTILAB® Refractometer
OPTILAB® Refractometer
Independent of chromaphores or fluorophores, differential Refractive Index (dRI) is an alternative concentration detector to UV-Vis spectrometer in all types of solvents. Based on each sample refractive index increment rather than the extinction coefficient to a given wavelength, it provides a complementary quantification method for MALS.
Specifications:
Range (RIU): -0.0047 to +0.0047
Peak-to-peak noise (RIU): 1 ± 7.5 x 10-10
Light Source: 660 nm
https://www.wyatt.com/products/instruments/optilab-refractive-index-detector.html
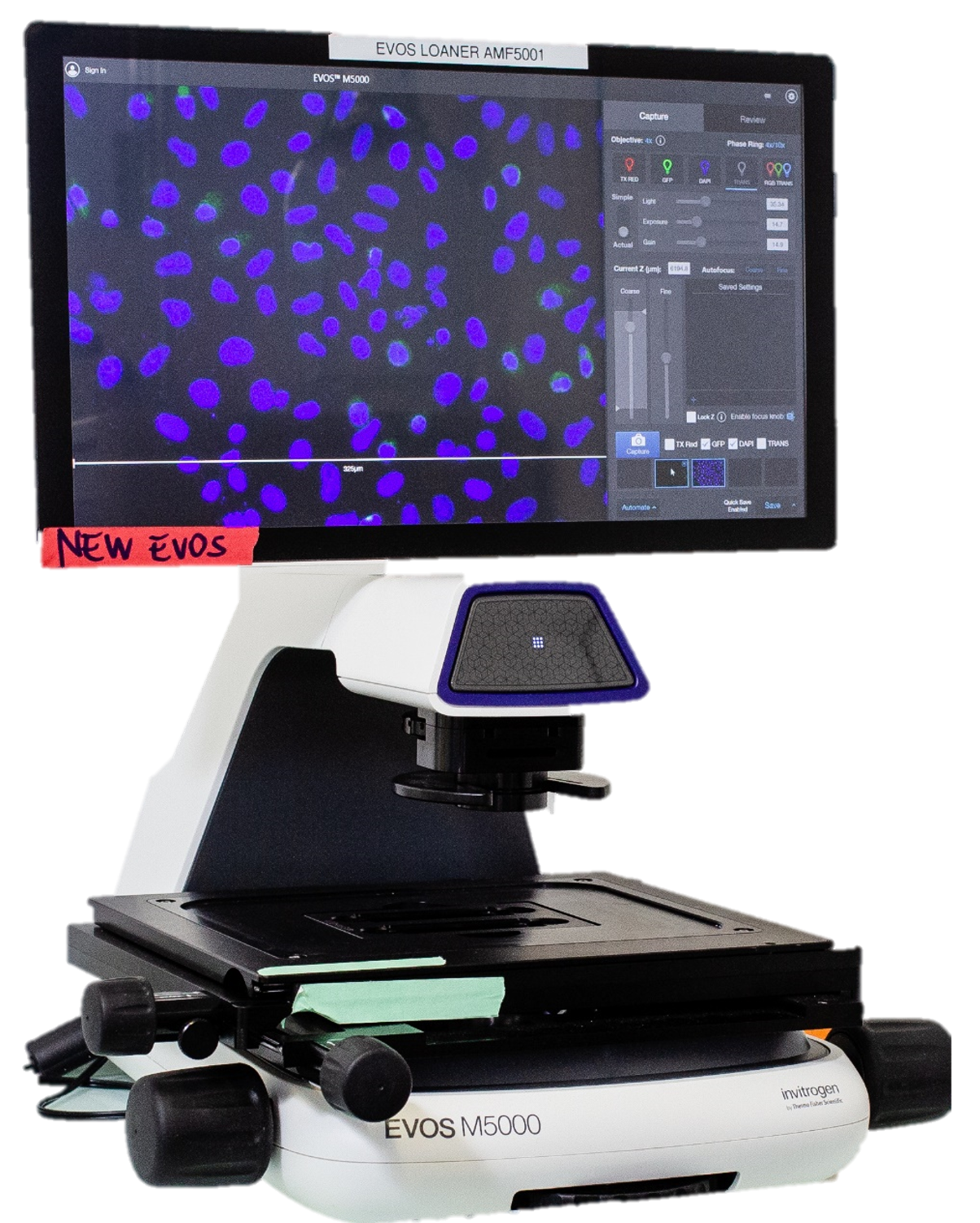
EVOS M5000 Imaging System – ThermoFisher
The EVOS M5000 is a four-color fluorescence, transmitted light imaging system. It features autofocus, Z-stack and time-lapse imaging capabilities.
The EVOS M5000 microscope combines an 18.5” high-resolution articulated LCD monitor, and a 3.2 MP monochrome CMOS camera (2048 x 1536) with 3.45-µm pixel resolution. The condenser includes a LED-based RGB illumination scheme that enables high-quality, low-noise color image acquisition using the same monochrome sensor, enabling rendering of true color in transmitted light.
The device features a five-position objective turret and the ability to acquire images in four fluorescence channels, transmitted light phase-contrast, and color.
Interchangeable vessel holders accommodate most vessel types and sizes, including slides, multi-well plates, culture flasks, and Petri dishes. Sample alignment is assisted by an automated stage.
Specifications:
Objectives: 4X, 10X, 20X, 40X.
Phase-contrast rings: 4X, 4X/10X, 20/40X
Output format: Monochrome RAW (16 bits): TIFF, PNG; Color (8 bits): TIFF, PNG, JPEG; Movies and stacks: AVI, WMV.
| Specifications | ||||
| Filters | Filter of excitation | Filter of emission | Dyes | Ref |
| DAPI | 357 nm | 447 nm | DAPI, Hoechst ™, BFP | AMEP 4950 |
| GFP | 482 nm | 524 nm | GFP, AlexaFluor™488, SYBR™, FITC | AMEP 4951 |
| TxRed | 585 nm | 628 nm | TexasRed™, AlexaFluor™568, AlexaFluor™594, MitoTracker™ Red, mCherry, Cy™3.5 | AMEP 4955 |

CellInsight - CX7-LZR - High content screening (HCS) – ThermoFisher
The CellInsight CX7 LZR platform is a system allowing simultaneously high content imaging and analysis thanks to the HCS Studio Cell Analysis software package capable of analyzing images in real time during their acquisition allowed by an automated confocal spinning-disk microscope.
Specifications:
The system has multiple technical features facilitating high-throughput image acquisition:
High-sensitivity camera: Photometrics™ X1 CCD cooled camera (2208*2208 px; 4,54 µm pixel size)
Objectives (Olympus) : 4x, 10x, 20x, 40x; automated turret with up to 3 objectives
Epi-illumination with 7-channel laser unit : 405, 450, 488, 561, 594, 647 and 785 nm
Colored brightfield imaging 4-channel LED array (447, 530, 590, 617 nm) for bright-field illumination allowing colorimetric absorbance measurements using traditional histology dyes
Multiplexing of up to five channels (near-UV to near-IR) allowed by a five-color dichroic emission filter system
Wide field and Confocal imaging : CrEST™ spinning disk unit equipped with 40 μm and 70 μm pinholes
Dual autofocus feature: far-red laser-based hardware autofocus and software autofocus algorithm
Plate scanning stage with interchangeable holders for multi-well plate (up to 1536 wells), 35 mm Petri dishes, microscopy slides, and 8-well chamber slides
Live cell imaging allowed by onstage incubator controlling temperature, humidity, and air mix composition (air, CO2, N2)
Software : HSC Studio, unique software for image acquisition and analysis. HSC Studio is a native CellInsight CX7 LZR software providing full control over the acquisition process along with multiple modes of automated image processing, statistical analysis, and data visualization. The software is capable of real-time analysis during acquisition.
Barcode reader for automated plate tracking.
SAFe 360 – Abbelight
SAFe 360 is an instrument for high resolution microscopy also called nanoscopy based on single fluorescent molecule localization microscopy (SMLM).
The SAFE 360 system commercialized by Abbeligth is mounted on an Olympus IX83 microscope.
This system allows to image and precisely localize a fluorescent molecule with a lateral localization accuracy of about 10 nm (at 100X magnification). Thanks to an astigmatic lens system, the system can reach a localization accuracy of 15 nm in z (at 100X magnification). The field of view could be large up to 150 x 150 mm2.
The system allows the recording of simultaneous multicolor imaging either in Epi-fluorescent but also in Total Internal Reflection Microscopy microscopy (TIRF) to study the thin region of interest (close to the coverslip, usually less than 200 nm). It is also possible to track the diffusion of single fluorescent molecule with a precision of 10 nm and a frame rate of approx. 20 msec/frame.
Specifications:
Excitation wavelengths: 405 nm (100 mW), 488 nm (100 mW), 561 nm (100 mW) and 640 nm (500 mW).
Spatial resolution (at 100X magnification): 10 nm in x and y and 15 nm in z
Sample types: fixed or live
Optimal sample size range: from µm to 10 nm
Imaging depth: up to 10 µm
https://www.abbelight.com/solutions/abbelight-instruments/#SAFe360


Raman microspectrometer DXR3xi – ThermoFisher
DXR3xi™ Raman confocal microscope allows imaging of biological content using molecular contrast. It is a laser-based technique triggering a light-matter interaction called Raman scattering. These scattering events occur with energetical shifts, of the excitation light, which are specific to the vibrational states of the molecules involved. By detecting the Raman scattered photons, it is possible to measure these energetical shifts and then estimate the spatially varying proportions of the molecules inside the sample. The DXR3xiTMalso includes a transmission microscope geometry combining brightfield/darkfield mode, with the possibility to switch quickly to one another by using a simple controller.
Specifications :
Ojectives: 10X, 20X, 50X (NA 0.75) and 60X water immersion
Motorized stage: positioning accuracy of 50 nm
Raman microscope
Excitation wavelengths: 532 and 785 nm.
Spatial resolution: ≈1 µm (at 50X magnification)
Spectral resolution: 5 cm-1 (2cm-1/pixel)
Spectral range: 50-3250 cm-1
Standard Microscope
Illumination modes: Brightfield / Darkfield
https://www.thermofisher.com/order/catalog/product/IQLAADGABFFAHCMBDI
QuickPlex SQ 120 MM – Meso Scale Discovery
The QuickPlex SQ 120 MM is a cutting-edge electrochemiluminescence technology developed by Meso Scale Discovery. This ultra-sensitive system enables the simultaneous detection of multiple biomarkers, providing rapid and precise analyses in various fields such as immunology, oncology, and infectious diseases.
The QuickPlex SQ 120 MM allows for the accurate measurement of proteins and other biomarkers in complex samples, including samples containing extracellular vesicles (secretome, biofluids, tissues).
To ensure an optimal experience, Meso Scale Discovery, in partnership with IVETh, offers personalized support with dedicated technical assistance and tailored training, facilitating the integration of this technology into your research projects.
https://www.mesoscale.com/en/products_and_services/instrumentation/quickplex_sq_120mm

